|
Unfortunately, there was a slightly different system in place in Europe. The modern atomic theory establishes the concepts of atoms and how they compose matter. Atoms are the ultimate building blocks of all matter. The periodic table is a way of organising the elements which is used by scientists to group elements with similar properties. To Learn how to Write Electronic Configurations, Detailed Explanation, Filling of orbital with FAQs, Visit BYJU’S for detailed explanation. Chemical elements are represented by a one- or two-letter symbol. In Electronic Configuration electrons are arranged in various shells, Subshell and Orbital by following certain rules. Atoms of these elements are characterized by having a partially filled 4f. While there is disagreement over exactly which elements should be included, many chemists state the lanthanides are elements with atomic numbers 58 through 71. The first two groups are 1A and 2A, while the last six groups are 3A through 8A. Electron Configuration -The Electron Configuration of an Element Describes how Electrons are Distributed in their Atomic Orbitals. The lanthanides are the elements in the top of the two rows located below the main body of the periodic table. They are assorted in a tabular arrangement wherein a row is a period and a column is a group. 
The traditional system used in the United States involves the use of the letters A and B. The periodic table is an arrangement of all the elements known to man in accordance with their increasing atomic number and recurring chemical properties. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. 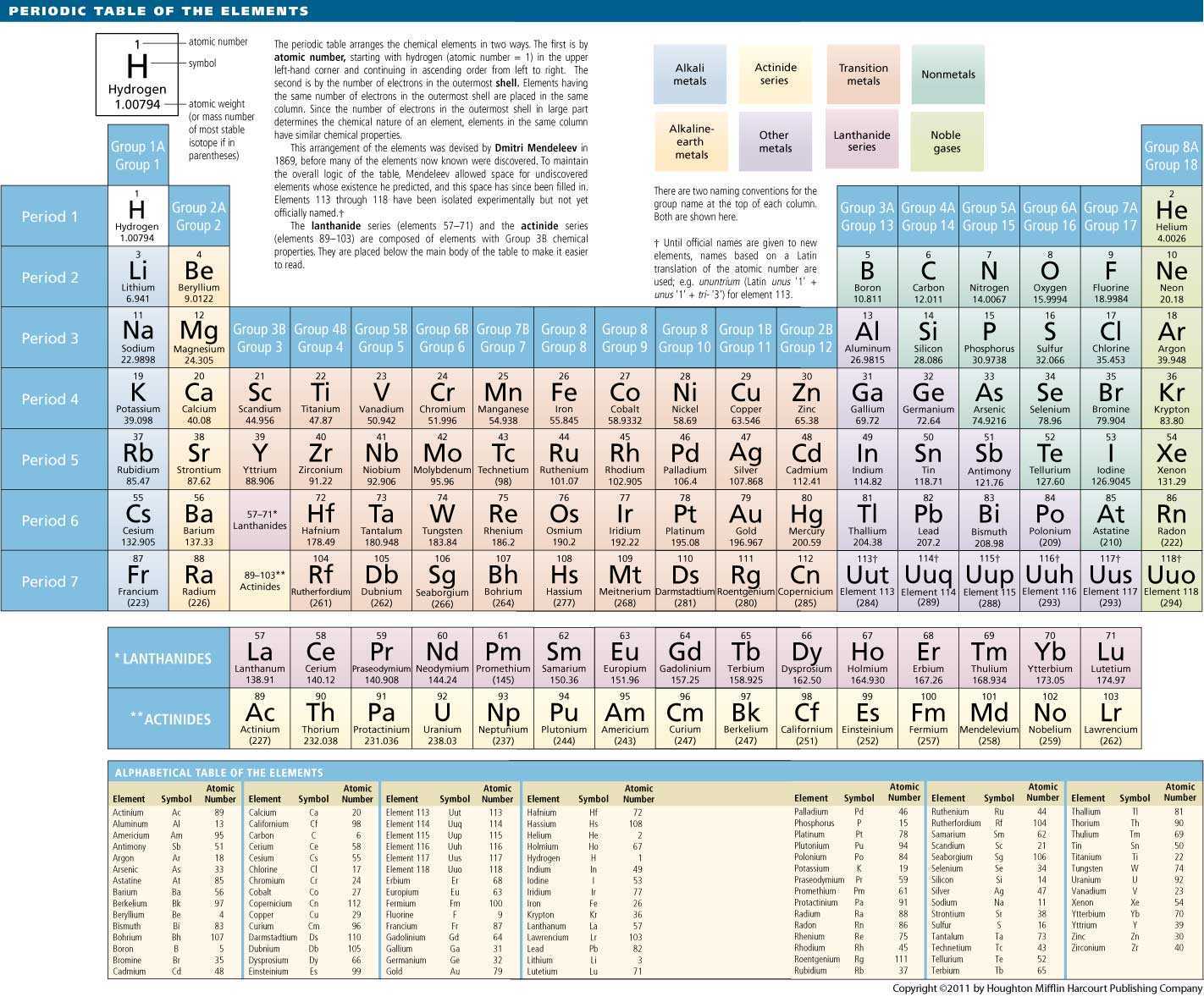
It is a physical science within the natural sciences that studies the chemical elements that make up matter and compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during reactions with other substances. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. Chemistry is the scientific study of the properties and behavior of matter. 
A new period begins when a new principal energy level begins filling with electrons. There are seven periods in the periodic table, with each one beginning at the far left. \) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
